创通网_配资评测网_全国前三股票配资_石家庄股票配资网/专业股票配资公司,是您值得信赖的投资伙伴,我们将始终陪伴您走过投资的每一个阶段,共同实现财富增长。
7月4日,英格卡购物中心携手瑞典国宝级I.... 07-14
为普及金融知识,提升保险行业服务品质,积。... 07-03
公元前 209 年美林资管,阳武县户牖乡。... 07-04
近日,北京第二外国语学院举办第十一届国际文化节,吸引了来自全球50多个国家和地区的1000余名国际学生参与。 国际文化节通过服装展示、歌舞表演等,展现了世界文化....
一句“我这个人脑子记不太清楚”,让张檬带娃去香港打疫苗的事瞬间冲上热搜。这番回应,不仅没翻车,反而因为其出人意料的“接地气”,引发了一场关于跨境医疗、育儿焦虑与....
亲爱的同学,如果你正在读物流管理专业或者考虑选择这个专业,你一定很关心未来的就业情况。我可以肯定地告诉你:大专物流管理专业不仅好找工作,而且前景十分广阔! 不过....
那天在超市货架前,我盯着琳琅满目的瓶装水,心里像压了块石头。儿子小宇刚满五岁,上周体检时医生轻声提醒:“孩子的钙摄入略低于标准,得多注意补充。”这句话像根刺,扎....
本周,锂电隔膜材料价格变动情况如下所示: 隔膜材料价格一览表(按周涨跌幅幅度排序) 品种周涨跌幅(%)月涨跌幅(%)日期价格/数量单位基膜(干法(16μm):国....
本周,酒类市场价格/产量变动情况如下所示: 国内酒类平均价格一览表(按周涨跌幅幅度排序) 品种周涨跌幅(%)月涨跌幅(%)日期价格/数量单位30年青花汾酒500....
《运输类旋翼航空器适航规定》已于2025年11月21日经第28次部务会议通过,现予公布,自2026年1月1日起施行。 部长 刘伟 2025年11月27日....
AI基金国联安精选混合(257020)披露2025年四季报盛宝优配,第四季度基金利润5675.15万元,加权平均基金份额本期利润0.0444元。报告期内,基金净....
AI基金汇添富社会责任混合A(470028)披露2025年四季报,第四季度基金利润148.64万元,加权平均基金份额本期利润0.0034元。报告期内九龙证券,基....
AI基金财通资管价值成长混合A(005680)披露2025年四季报,第四季度基金利润2969.14万元,加权平均基金份额本期利润0.049元。报告期内,基金净值....
 创通网文章加载中,请稍后...
创通网文章加载中,请稍后...
一句“我这个人脑子记不太清楚”,让张檬带娃去香港打疫苗的事瞬间冲上热搜。这番回应,不仅没翻车,反而因为其出人意料的“接地气”,引发了一场关于跨境医疗、育儿焦虑与....
亲爱的同学,如果你正在读物流管理专业或者考虑选择这个专业,你一定很关心未来的就业情况。我可以肯定地告诉你:大专物流管理专业不仅好找工作,而且前景十分广阔! 不过....
近日,北京第二外国语学院举办第十一届国际文化节,吸引了来自全球50多个国家和地区的1000余名国际学生参与。 国际文化节通过服装展示、歌舞表演等,展现了世界文化....
那天在超市货架前,我盯着琳琅满目的瓶装水,心里像压了块石头。儿子小宇刚满五岁,上周体检时医生轻声提醒:“孩子的钙摄入略低于标准,得多注意补充。”这句话像根刺,扎....
《运输类旋翼航空器适航规定》已于2025年11月21日经第28次部务会议通过,现予公布,自2026年1月1日起施行。 部长 刘伟 2025年11月27日....
本周,锂电隔膜材料价格变动情况如下所示: 隔膜材料价格一览表(按周涨跌幅幅度排序) 品种周涨跌幅(%)月涨跌幅(%)日期价格/数量单位基膜(干法(16μm):国....
本周,酒类市场价格/产量变动情况如下所示: 国内酒类平均价格一览表(按周涨跌幅幅度排序) 品种周涨跌幅(%)月涨跌幅(%)日期价格/数量单位30年青花汾酒500....
AI基金国联安精选混合(257020)披露2025年四季报盛宝优配,第四季度基金利润5675.15万元,加权平均基金份额本期利润0.0444元。报告期内,基金净....
AI基金财通资管价值成长混合A(005680)披露2025年四季报,第四季度基金利润2969.14万元,加权平均基金份额本期利润0.049元。报告期内,基金净值....
AI基金汇添富优选价值混合发起式A(021198)披露2025年四季报,第四季度基金利润93.55万元,加权平均基金份额本期利润0.0673元。报告期内,基金净....
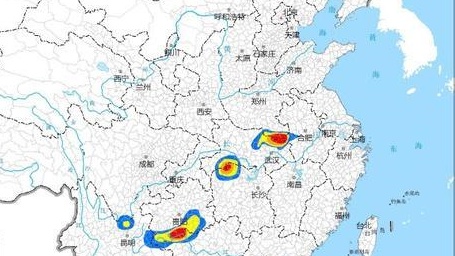
据央视新闻,根据河南省气象台监测显示,6月30日6时至7月1日6时,河南省北中部、西部出现阵雨、雷阵雨,其中,鹤壁、新乡、三门峡、洛阳、平顶山、许昌、南阳等地部....
7月4日,英格卡购物中心携手瑞典国宝级IP《长袜子皮皮》打造的“宜起‘皮’一夏”夏日玩乐季正式启幕,首站落地北京荟聚。据介绍领航配资,英格卡购物中心隶属于英格卡....
为普及金融知识,提升保险行业服务品质,积极履行社会责任,近期赢翻网配资,中华财险广州中心支公司组织开展“7.8全国保险公众宣传日”系列宣传活动。 营造浓厚氛围,....
公元前 209 年美林资管,阳武县户牖乡的社祭现场。一个身材高大、面容俊朗的青年正握着屠刀,有条不紊地分割祭肉。他的手法精准如尺,每一块肉都厚薄均匀、分量相等,....
本站消息龙辉配资龙辉配资,根据天眼查APP显示,近日公布了一则上海哈啰普惠科技有限公司作为被告/被上诉人的开庭公告,详细内容如下: 案号:(2025)浙0212....
一家LED灯具企业在出口申报时,将产品归类填错了,产品运到国外后,被当地海关重罚了9万美元,如果提前做好功课,这种情况完全可以避免6月26日下午2点,江苏苏州高....
为更好向社会展示监狱对党忠诚、依法治监、深耕改造的良好形象,6月25日,上海市四岔河监狱邀请民主党派、执法监督员、检察院检察官、民警家属代表、服刑人员亲属、新闻....
德国总理默茨会见王毅双盈策略 当地时间2025年7月4日,德国总理默茨在柏林会见中共中央政治局委员、外交部长王毅。 默茨表示,德中关系发展良好双盈策略,政治经贸....
7月2日,以“深挖大数据价值激发数字经济新动能”为主题的2025全球数字经济大会大数据创新应用论坛在北京国家会议中心召开。据悉,论坛上,发布了五项重要内容,贯通....
收缴率低、长期亏损,很多物业公司选择不干了。 6月25日,中海物业向鄂州双创之星小区发出撤场公告信,表示公司将于2025年8月31日18:00正式退出项目,结束....
